General Chemistry
Prices subject to change without prior notice.
Click on an image to enlarge.
Individual Kits
| Kit Name | Part # INSTRUCTIONS/LAB ACTIVITES | Description | Price US $ |
OUCH (Original Ultimate Comprehensive Heuristic) Chemistry Modeling Kit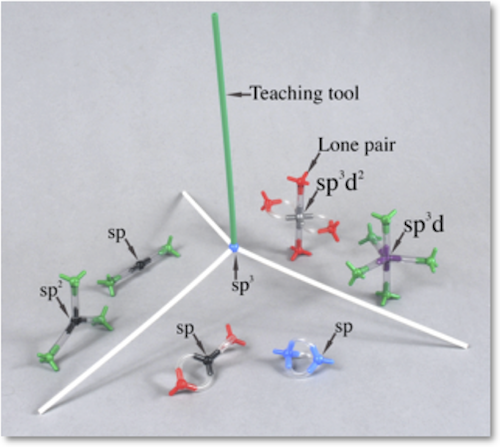  | OUCH-1 | A Complete Teaching and Learning Kit for Modeling: Covalent Bonding VSEPR Theory Balancing Equations Enthalpy Change Equilibrium Acid-Base Reactions Reaction Mechanisms Dispersion Forces Hydrogen Bonding Water and Ice Solvation σ and π Bonds Functional Groups Isomers Reaction Quotients Kit Contents: 30 black 4-peg carbon atom centers 15 black 3-peg boron atom centers 90 white 1-peg hydrogen atom centers 60 red 4-peg oxygen atom centers 16 yellow 6-peg metal or sulfur atom centers 15 yellow 4-peg sulfur atom centers 30 blue 4-peg nitrogen atom centers 90 green 4-peg halogen atom centers 16 purple 5-peg phosphorous atom centers 15 black 2-peg beryllium atom centers 17 21cm Straws 90-1.25" PVC, clear and colorless bonds (single bonds) 90-4cm PVC, clear bonds and colorless (double bonds) 16-0.87" clear and colorless PVC bonds 14-0.87" white, PVC bonds 6-0.87" black, PVC bonds 6-2.12" white, PVC bonds (pi bonds) 30 Magnets | $185.40 |
GENERAL CHEMISTRY | GEN-1 | Kit Contents: 12 black 4-peg carbon atom centers 12 black 3-peg carbon atom centers 20 white 1-peg hydrogen atom centers 6 red 2-peg oxygen atom centers 3 red 4-peg oxygen atom centers 8 yellow 4-peg sulfur atom centers 3 blue 4-peg nitrogen atom centers 6 green 4-peg halogen atom centers 6 green 1-peg halogen atom centers 3 light green 1-peg halogen atom centers 3 dark green 1-peg halogen atom centers 4 purple 5-peg phosphorous atom centers 2 silver 6-peg sulfur (xenon) atom centers 50-1.25" PVC, clear bonds (single bonds) 2-0.75" PVC, clear bonds (sigma bonds) 12-1.625" PVC, clear bonds (double bonds) 4-1.625" white, PVC bonds (pi bonds) Background information Model building activity PDF on this website. The background information topics include ionic and covalent bonding, orbital hybridization, and sigma and pi bonding. Several inorganic and organic compounds are suggested for building, using this kit. | $29.30 |
VSEPR I (BEGINNING SET) | VSP-1 | Kit Contents: 5 white 1-peg hydrogen atom centers 3 blue 4-peg nitrogen atom centers 4 red 4-peg oxygen atom centers 3 green 4-peg halogen atom centers 2 black 4-peg carbon atom centers 2 yellow 4-peg sulfur atom centers 8-1.25" PVC, clear bonds (single bonds) 8-1.625" PVC, clear bonds (double bonds) Background information booklet, Lab procedure, student response form, and answer key PDF on this website. The accompanying booklet explains the geometry of valence electron pairs, based on VSEPR theory, and molecular shape using ABE notation. Instructions are given for drawing correct Lewis structures of several molecules which can then be built with this kit. The lab procedure calls for students to make examples of linear, bent triatomic, tetrahedral, and trigonal planar molecules. | $8.65 |
VSEPR I CLASSROOM SET | VSP-1C | Classroom Set Contents: 75 white 1-peg hydrogen atom centers 45 blue 4-peg nitrogen atom centers 60 red 4-peg oxygen atom centers 45 green 4-peg halogen atom centers 30 black 4-peg carbon atom centers 30 yellow 4-peg sulfur atom centers 120-1.25" PVC, clear bonds (single bonds) 120-1.625" PVC, clear bonds (double bonds) 1 Sturdy storage box Background information booklet Lab procedure, student response form, and answer key The classroom set has enough parts for 15 pairs of students to do the lab procedure. The accompanying booklet explains the geometry of valence electron pairs, based on VSEPR theory, and molecular shape using ABE notation. Instructions are given for drawing correct Lewis structures of several molecules which can then be built with this kit. The lab procedure calls for students to make examples of linear, bent triatomic, tetrahedral, and trigonal planar molecules. | $104.45 |
| VSEPR II (ADVANCED SET) | VSP-2 | Kit Contents: 4 red 4-peg oxygen atom centers 7 green 4-peg halogen atom centers 2 purple 5-peg phosphorous atom centers 2 silver 6-peg sulfur (xenon) atom centers 2 yellow 4-peg sulfur atom centers 12-1.25" PVC, clear bonds (single bonds) 12-1.625" PVC, clear bonds (double bonds) Background information booklet Lab procedure, student response form, and answer key As in the VSEPR I kit, the accompanying booklet explains the geometry of valence electron pairs, based on VSEPR theory, and molecular shape using ABE notation. Instructions are given for drawing correct Lewis structures of several molecules which can then be built with this kit. In addition, the more advanced topic of orbital hybridization is covered. The lab procedure for this kit gives students hands-on experience making models with linear, trigonal bipyramidal, T-, square pyramidal, and see-saw shapes. | $10.10 |
VSEPR II CLASSROOM SET | VSP-2C | Kit Contents: 60 red 4-peg oxygen atom centers 105 green 4-peg halogen atom centers 30 purple 5-peg phosphorous atom centers 30 silver 6-peg sulfur (xenon) atom centers 30 yellow 4-peg sulfur atom centers 180-1.25" PVC, clear bonds (single bonds) 180-1.625" PVC, clear bonds (double bonds) Background information, lab procedure, student response form, and answer key available as a PDF. As in the VSEPR I kit, the accompanying PDF explains the geometry of valence electron pairs, based on VSEPR theory, and molecular shape using ABE notation. Instructions are given for drawing correct Lewis structures of several molecules which can then be built with this kit. In addition, the more advanced topic of orbital hybridization is covered. The lab procedure for this kit gives students hands-on experience making models with linear, trigonal bipyramidal, T-, square pyramidal, and see-saw shapes. | $126.55 |
CHEMISTRY OF WATER | WAT-1 | Kit Contents: 12 red 4-peg oxygen atom centers 1 silver 6-peg sodium ion 1 green 6-peg chloride ion 36-0.75" PVC, clear tubes (lone pairs of electrons) 24-0.75" PVC, white tubes (hydrogen atoms) 60-4mm x 1/2" cylindrical magnets Assembly instructions Background information booklet Lab procedure, student response form, and answer key Each water molecule has a central 4-peg, red-oxygen atom center to which are bonded two white tubes representing the hydrogen atoms and two clear tubes to show the two pairs of nonbonding electrons of the oxygen atom. Cylindrical, very strong magnets are aligned within the four tubes so that the free ends of the two hydrogen atoms are south, while the lone electron pair ends are both north. Therefore, the hydrogen of one molecule bonds to a lone pair of electrons on another water molecule. The south ends of the magnets placed in the clear tubes on the sodium ion will attract the water's lone pairs, and the north ends of the chloride ion will bond to the hydrogens of the water. Science classes from 5th grade through high school will benefit from investigating the properties of this most ubiquitous substance. The kit demonstrates hydrogen bonding, molecular shape, bond and molecular polarity, expansion of water on cooling, solvation of a salt, surface tension, tension and cohesion, transpiration in plants, colligative properties of water, etc. | $31.70 |
COORDINATION CHEMISTRY | COR-1 | Kit Contents: 5 silver 6-peg metal atom centers 11 blue 4-peg nitrogen atom centers 41 white 1-peg hydrogen atom centers 17 red 4-peg oxygen atom centers 8 green 1-peg halogen atom centers 12 black 4-peg carbon atom centers 70-1.25" PVC, clear bonds (single bonds) 13-1.625" PVC, clear bonds (double bonds) Background information booklet Lab procedure, student response form, and answer key Topics covered in the information booklet include composition and naming of coordination compounds, ligand classification ( mono-, bi-, and polydentate), and stereoisomerism. In the lab procedure, students fill in response sheets and build models of ligands and complex ions. Models are used to determine ligand classification and type of isomerism. | $31.40 |
ICE | ICE-1 | Kit Contents: 50 red 4-peg oxygen atom centers 100 white 2-peg hydrogen atom centers 74-0.75" PVC, clear bonds (hydrogen bonds) 100-0.75" PVC, white bonds (covalent bonds) Assembly instructions and Lab procedures PDF on this website. Two layers of hydrogen- bonded water molecules show the crystalline, open structure of ice. | $47.80 |
ICE | ICE-2 | Kit Contents: 50 red 4-peg oxygen atom centers 98 white tubes for hydrogen atoms 98 rigid, grey tubes for hydrogen bonds Assembly instructions and Lab procedures PDF on this website. This model is more compact than ICE-1, but also has two layers of hydrogen-bonded water molecules showing the crystalline, open structure of ice. | $32.65 |
BOND ANGLE DETERMINATION | BAD-1 | Kit Contents: 9 red cube corner vertices 2 green tetrahedral atom center 4 red 21cm long tubes 2 green 21cm long tubes Lab activity, and teacher's guide Integrating chemistry, math (trigonometry, geometry), logic, and problem solving, this model challenges students to determine the bond angles of a tetrahedral atom center. | $9.75 |
| TETRAHEDRAL BOND ANGLE DETERMINATION CLASSROOM SET | BAD-1C | Kit Contents: 122 red cube corner vertices 17 green tetrahedral atom centers 60 red 21cm long tubes 30 green 21cm long tubes Lab activity, and teacher's guide The classroom set has enough parts for 15 pairs of students to do the lab procedure. Integrating chemistry, math (trigonometry, geometry), logic, and problem solving, this model challenges students to determine the bond angles of a tetrahedral atom center. | $120.90 |
